
PubChem is a reliable source of chemical information from authoritative sources. In Group 1, the reactivity of the elements increases going down the group. Students should be able to describe the reactions of the first three alkali metals with oxygen, chlorine and water. 
You can look up element names, symbols, atomic masses and more, or test your knowledge with a periodic table game. 5.1 Atomic structure and the periodic table. They have comparable characteristics because the number of valence shell electrons is the same. PubChem Periodic Table of Elements is an interactive tool that lets you explore the properties and trends of chemical elements. The electrical configuration of the valence shell of the elements is repeated in such a manner that elements with the same valence shell electrons fall under the same group when they are arranged in order of increasing atomic number. The electrical configuration of the elements provides the basis for the current periodic table. Periodic Table Trends (size & reactivity) quiz for 9th grade students. The higher the electronegativity, the farther right and up on the periodic table you go. As you move from left to right across a row on the periodic table atoms tend to get smaller because. The stronger the electronegativity, the more intense the electron exchange. Stile Education 10.3K subscribers Subscribe Subscribed 2.3K 150K views 3 years ago Chemistry In this short series, we look at what makes certain elements really reactive and others just.not. Over the course of a time, reactivity rises as you move from left to right.Īs you move down the group, your reactivity declines. Periodic trends (such as electronegativity, electron affinity, atomic and ionic radii, and ionization energy) can be understood in terms of Coulombs law, which is F (qq)/r². The simpler it is for electrons to be given or taken away the farther to the left and down the periodic chart you travel, the greater the reactivity. Period - As you go from left to right throughout a period, your responsiveness diminishes.Īs you go down a group, your reactivity rises. Because the transfer/interaction of electrons is the foundation of chemical processes, this is generally defined by how readily electrons can be withdrawn (ionisation energy) and how strongly they desire to take other atom's electrons (electronegativity). The transition metals, groups 312 in the periodic table, are generally characterized by partially filled d subshells in the free elements or their cations. The reactivity of an atom relates to how probable it is for it to react with other substances. To understand the trends in properties and reactivity of the d-block elements. That is why elements in the periodic table have periodic physical and chemical characteristics. YouTube 0:00 / 3:37 Intro Trends in the Periodic Table Reactivity Stile Education 10.3K subscribers Subscribe Subscribed 2. Elements are placed in the contemporary periodic table according to their atomic numbers, which, as previously established, are closely connected to their physical and chemical characteristics. This video also goes through some notes on reactivity trends.Hint: “Physical and chemical characteristics of the elements are periodic functions of their atomic numbers,” according to Moseley's Modern Periodic Law. This video will go through patterns of reactivity for group 1. a diagonal division on the periodic table that divides the metals from the nonmetals and along which lay the metalloids. a family of non-reactive gases (family 18) on the periodic table. Reactivity generally increases from the centre of the periodic table to the left (recall the right-hand side has non-metals), and also from the top of the periodic table down. a family within the nonmetals (family 17) on the periodic table. See if you can apply your knowledge of the ionisation energy (energy required to expel an electron from an atom), electronegativity (ability to attract electrons to atom) and atomic radii (half the distance between two neighbouring nuclei of the same element) trends on the periodic table. This video will go through some general reactivity trends across the periodic table. The specific arrangement of Group 1 elements on the periodic table allows us to make predictions about their reactivity. 
IE increases bc the radius decreases, electrons feel the pull of the nucleus more. IE decreases bc the atomic radius increases, valence electrons dont feel the pull of the nucleus as much. Analyse patterns in metal activity on the periodic table and explain why they correlate with, for example: the amount of energy required to remove an electron.This post will go through some patterns in metal activity for Prelim Chemistry. 45 Reactivity of Elements of the Periodic Table Novice 1 m Reactivity is a measure of how easily an element will combine with other elements to form compounds. 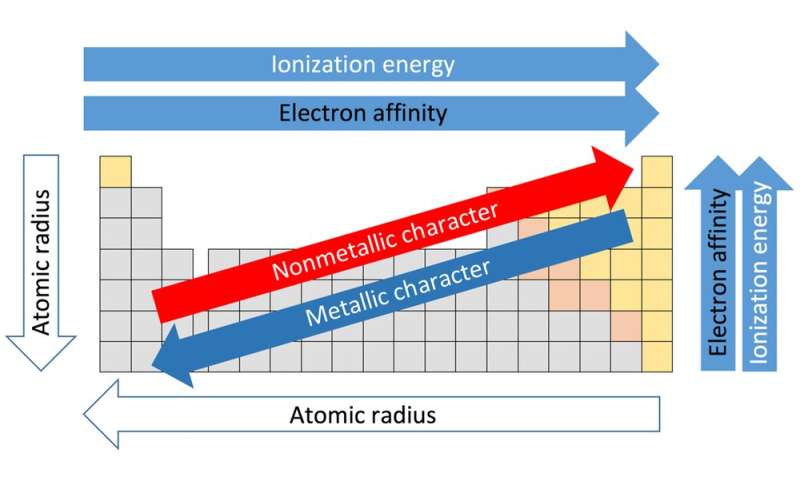
What Are Some Patterns in Metal Activity?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
